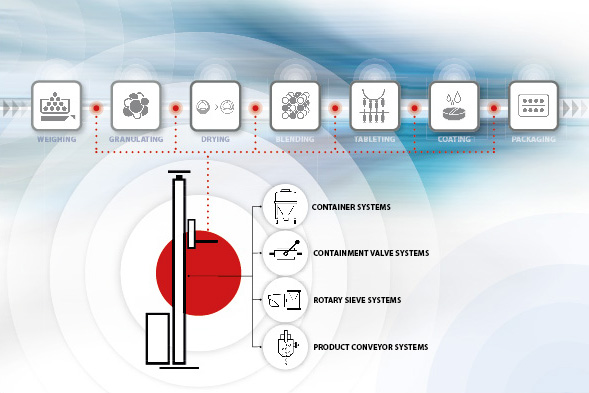
From weighing out to mixing, sieving, granulating, pressing into tablets, coating, and packing, it is clear just how many steps are involved in processing and producing solid pharmaceuticals. Manufacturers generally demand a great deal from individual machines, requiring the processes that they perform to be the best in their class. But the question of how the product should get from process A to process B is often given less consideration. “This is a mistake that can have dire consequences,” says Erich Nussbaumer, an employee at pharmaceutical specialist Glatt. Nussbaumer, a material handling expert, is making the case for integrated handling – from the moment the materials are received through to the fully packed dosage form and on to shipping the finished product. It is only when the entire process chain is carefully considered and seamlessly realized that certain requirements can be met – such as flexible and easily modified systems with a high degree of automation. A trend that is currently on the rise supports this way of thinking: Transfer and handling systems need to be sophisticated and carefully designed in order to manage the growing volumes of highly potent active ingredients that they are being faced with. This means that equipment such as containment valve systems must be built in a way that enables them to be used in as many different processes as possible. The same applies to cleaning systems and lifting devices. As well as flexibility and compatibility, safety is a key consideration when it comes to handling toxic substances.
As Nussbaumer explains: “Automation is becoming increasingly important of ensuring process safety, and protection of personnel in particular, when processing new and highly active ingredients. Loading, sample taking, discharging, and cleaning are the most important process steps, as the possibility of handling errors occurring during any of them has to be ruled out altogether.” Automation is a useful tool for achieving this, too. In order for it to play this role, the system components that work together must be connected intelligently through digitalization methods so that any errors can be eliminated as a result. At the moment, the system supplier is specifically working on a system in which RFID tags on product containers and handling systems enable products to be tracked automatically across the entire process chain, preventing handling errors.
Flexible Systems Require a Sophisticated Material Flow
This trend in favor of flexible systems is also being fueled by the structural changes taking place in the pharmaceutical industry across the globe. Axel Friese, Head of Marketing at Glatt, attests to this: “While Asia is ramping up its production of large batches, Europe is increasingly expressing a need for flexible systems that can be used for alternating products and small batches.” Manufacturers are using their standard processes as a basis for developing full process chains that operate in perfect harmony. To make new solutions – such as integrating robots – a reality in projects like these, companies are turning to the expertise of the staff members at the subsidiary Glatt Ingenieurtechnik. Material handling poses particularly intricate challenges in cases where highly potent active ingredients need to be processed under high containment conditions. “A key element is the interplay between our TKS containment valve system and the handling equipment,” explains Erich Nussbaumer. When this was established, the main challenge was to maintain a closed, contamination-free product flow with entirely dust-free loading and dispensing, and to separate processes in a way that would avoid introducing any contaminants at all. This is where the divided TKS containment valve system from Glatt came in. “Generally, we always offer two solutions: either system configurations in which the operator uses full protection gear or containment systems that do not require full protection gear. But we are seeing a clear move away from manual or hybrid operation of pharmaceutical systems – fully automated and closed processes are the future,” explains Nussbaumer.
The company’s current technical developments in material handling include RFID tracking and developing modular systems with a focus on integrating and monitoring manual process steps. As Axel Friese states: “We are seeing more of a demand for process safety and reliability, so manufacturers and designers are giving more consideration to material handling as a result.”






 Copyright: Fachwelt Verlag
Copyright: Fachwelt Verlag